Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Baba, Yuji; Shimoyama, Iwao; Hirao, Norie; Izumi, Toshinori
e-Journal of Surface Science and Nanotechnology (Internet), 13, p.417 - 421, 2015/09
Times Cited Count:1The interaction between alkali metals and oxides has attracted much attention as heterogeneous catalysis, chemical reaction promoters, and high intensity electron emitter. Also the interaction of cesium and oxides has become important subject, because radioactive cesium trapped in minerals such as clay and soil may cause health risks. In the present study, we analyzed chemical states of ultra-trace amount of cesium on oxide surfaces by total reflection X-ray photoelectron spectroscopy (TR-XPS) exited by synchrotron radiation. For the adsorbed cesium thicker than 0.01 layer, cesium is weakly bound with oxide through Van-der-Waals force. On the other hand, for ultra-thin layer about 0.002 layer, the chemical bond changes to covalent bond. It is suggested that this change in the chemical bonding state is one of the reasons why radioactive cesium is hard to be released from minerals.
Nagase, Kenzo*; Morita, Noboru*; Watanabe, Akiyoshi*; Asao, Toyonobu*
JAERI-Tech 2005-052, 99 Pages, 2005/09
no abstracts in English
Morimoto, Yoshiki*; Kato, Sayuri*; Toda, Naohiro*; Katayama, Yoshinori; Tsuji, Kazuhiko*; Yaoita, Kenichi*; Shimomura, Osamu
Review of High Pressure Science and Technology, 7, p.245 - 247, 1998/03
X-ray diffraction for liquid Rb and liquid Na has been measured under pressure up to 6 GPa using synchrotron radiation. Volume dependences of static structure factor 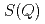 and pair distribution function
and pair distribution function 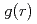 were obtained to the volume range
were obtained to the volume range 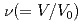 of 0.52 for liquid Rb and
of 0.52 for liquid Rb and 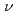 of 0.73 for liquid Na to investigate the effect of the electronic change on the structure of liquid metals. With increasing pressure, the peaks of
of 0.73 for liquid Na to investigate the effect of the electronic change on the structure of liquid metals. With increasing pressure, the peaks of 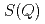 of both liquid Rb and liquid Na shift towards higher
of both liquid Rb and liquid Na shift towards higher  , and the heights of the first peak increase. Theses volume dependences of the structural data are compared with those for other metals under pressure and expanded fluids.
, and the heights of the first peak increase. Theses volume dependences of the structural data are compared with those for other metals under pressure and expanded fluids.
; Chihara, Junzo
Molecular Simulation, 16, p.31 - 46, 1996/00
Times Cited Count:4 Percentile:17.3(Chemistry, Physical)no abstracts in English
 UF
UF and Rb
and Rb UF
UF
; ; ; Fujino, Takeo
Inorg.Chim.Acta, 54, p.L193 - L194, 1981/00
no abstracts in English
Furukawa, Kazuo
Oyo Butsuri, 44(8), P. 877, 1975/08
no abstracts in English
Furukawa, Kazuo;
Bussei Kenkyu, 19(5), p.B35 - B45, 1973/05
no abstracts in English
Furukawa, Kazuo
Kosoku Denjiekiryu Ni Kansuru Chosa Hokokusho, p.159 - 170, 1973/00
no abstracts in English